Clinical Development & Execution Support for Life Sciences
Senior clinical leadership and hands on execution when your study requires clarity, structure, and steady direction.
CIMI supports device, diagnostics, and life science companies at critical stages of clinical development. When study design needs refinement, documentation must meet higher regulatory standards, or experienced oversight is required to stabilize execution, we provide focused clinical leadership without expanding internal infrastructure.
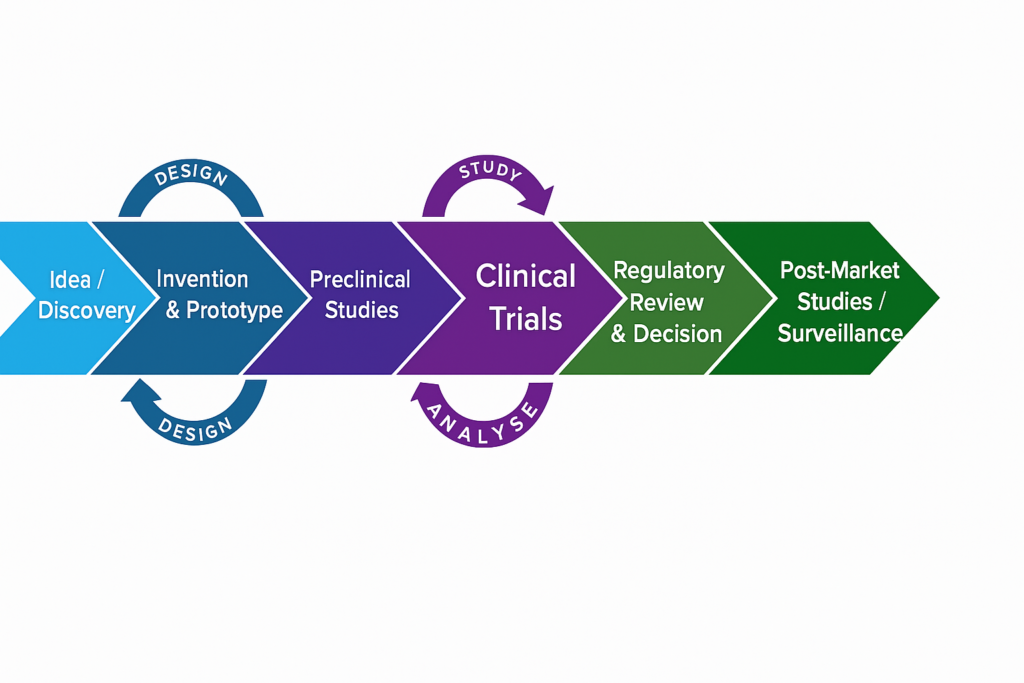
Clinical Strategy & Study Design
Translate scientific and business objectives into structured, regulator ready clinical programs that are realistic, defensible, and execution focused.
Examples include:
• Study strategy development
• Protocol concept and endpoint alignment
• Regulatory ready study design
• Risk identification and mitigation planning

Protocol & Essential Document Development
Develop high quality clinical documentation that meets regulatory expectations and supports operational success.
Examples include:
• Protocol development and revision
• CRFs and study tools
• Informed consent and essential documents
• Study manuals and supporting materials
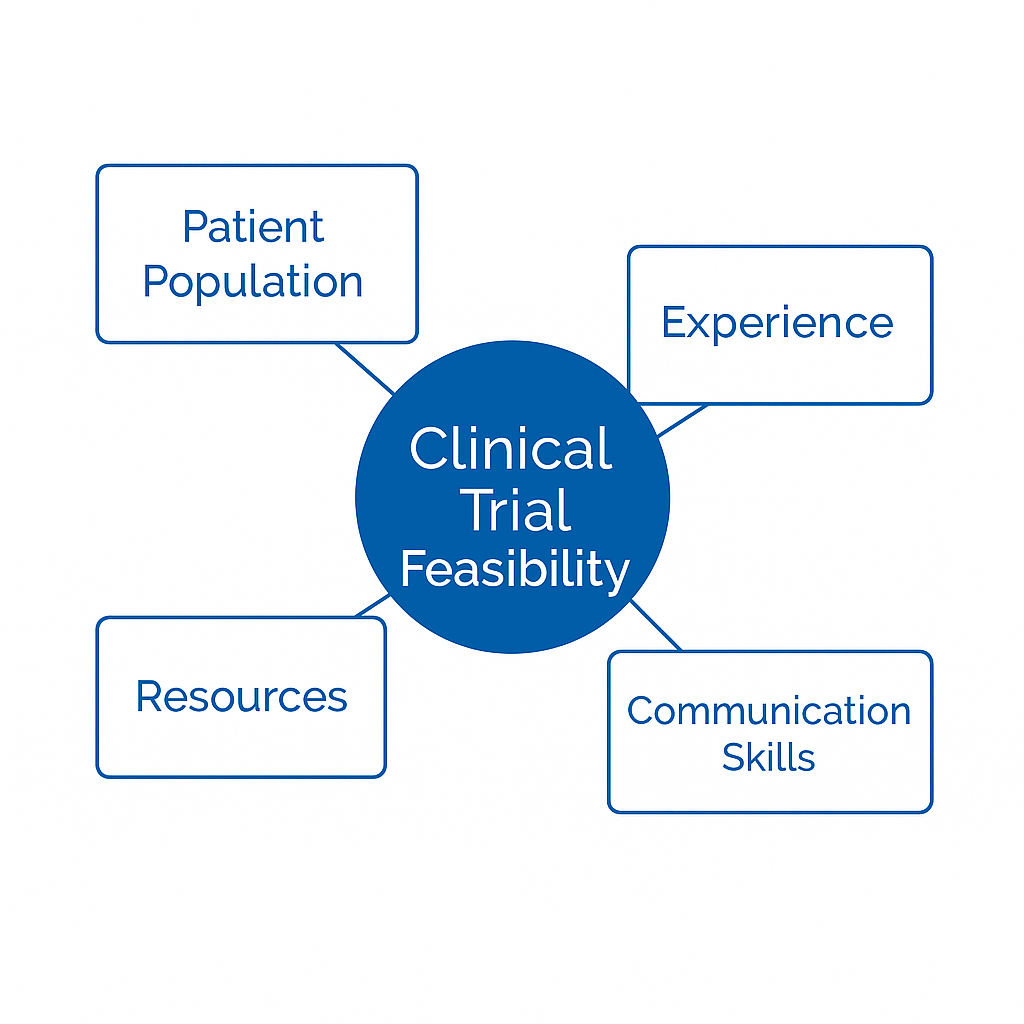
Clinical Project Leadership Support
Provide experienced clinical leadership to guide decision making and maintain momentum across the study lifecycle.
Examples include:
• Interim clinical leadership support
• Study planning and execution oversight
• Cross functional coordination
• Timeline and milestone management

Targeted Monitoring & Oversight
Provide experienced clinical oversight that strengthens data integrity and supports compliant, consistent study conduct.
Examples include:
• Risk based monitoring strategy
• On site and remote monitoring support
• Source data review and issue resolution
• Audit and inspection readiness support
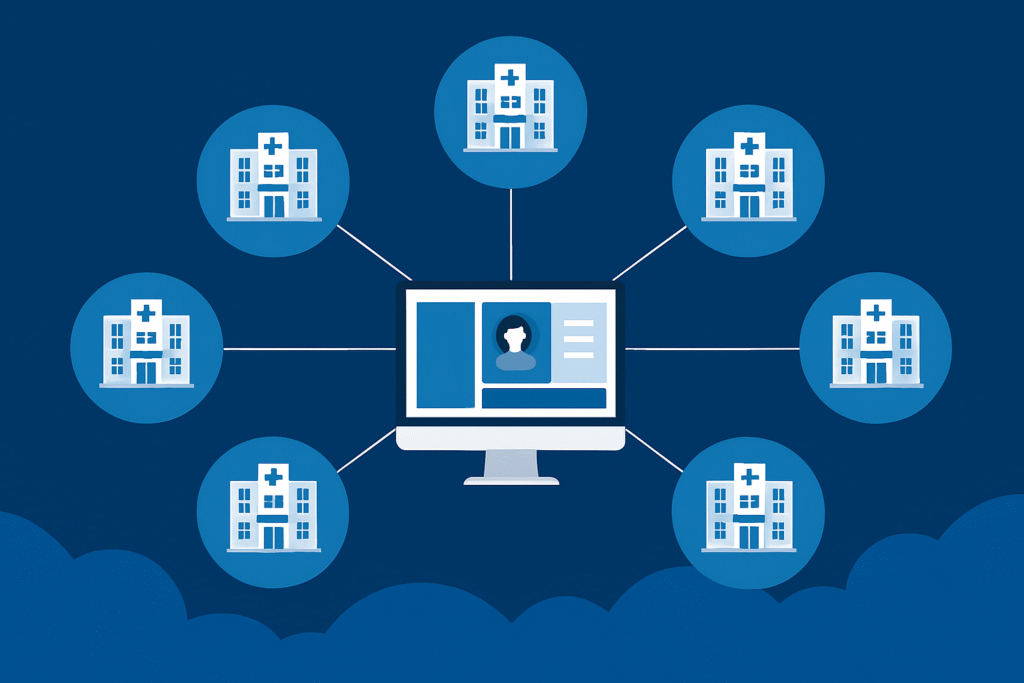
Site & Investigator Enablement
Equip investigators and site teams with the structure and guidance needed to execute confidently and consistently.
Examples include:
• Investigator training and study start up support
• Site initiation preparation
• Study conduct guidance and communication
• Ongoing site engagement and issue management

Data Integrity & Quality Perspective
Apply senior clinical judgment to identify risks early and protect the credibility of study outcomes.
Examples include:
• Data review and trend analysis
• Protocol deviation assessment
• CAPA support and documentation review
• Quality oversight aligned with regulatory expectations
Advance Your Product Toward Submission with Confidence
Clinical design, documentation, and oversight shape the strength of your regulatory package. CIMI provides experienced clinical leadership to ensure your study supports a clear and defensible path forward.
Schedule a strategic clinical discussion.